50+ Sample Balance Sheets
-

Sample Balance Sheet Template
download now -

Understanding Balance Sheet
download now -

Annual Balance Sheet
download now -
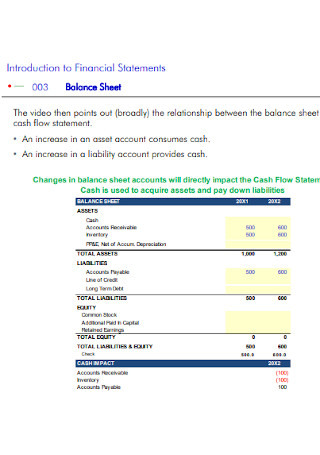
Financial Balance Sheet Template
download now -
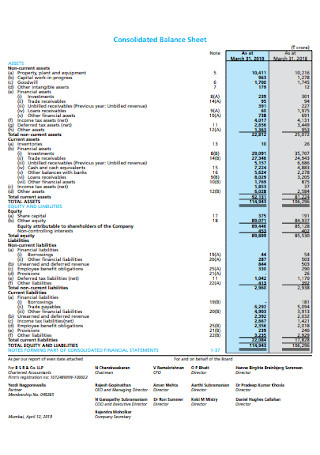
Consolidated Balance Sheet
download now -
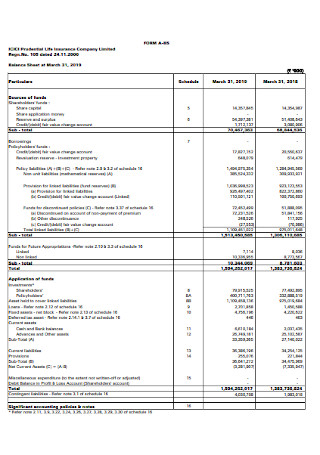
Insurance Company Balance Sheet
download now -

Profit and Loss Balance Sheet
download now -

Bank Balance Sheets
download now -
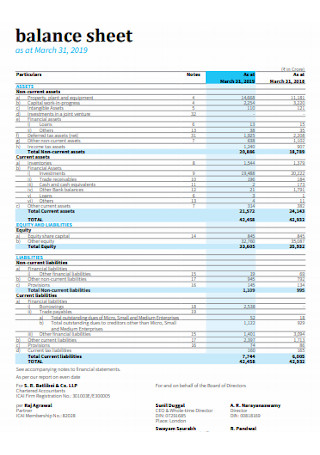
Simple Balance Sheet Template
download now -
Balance Sheet of Cash Flow Statement
download now -
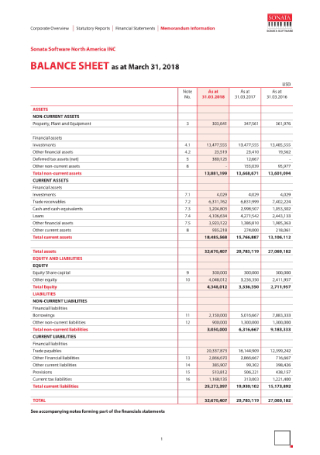
Company Balance Sheet Example
download now -

House Balance Sheet
download now -
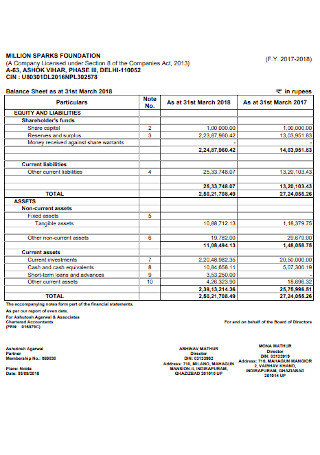
Foundation Balance Sheet
download now -

Annual Balance Sheet Report
download now -
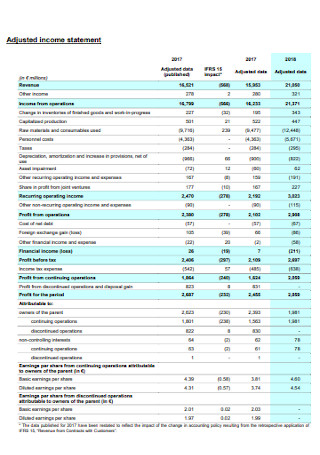
Income and Balance Sheet
download now -
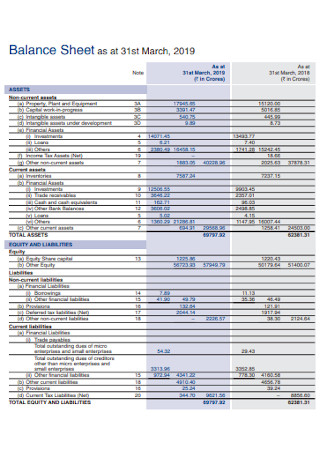
Year Balance Sheet
download now -
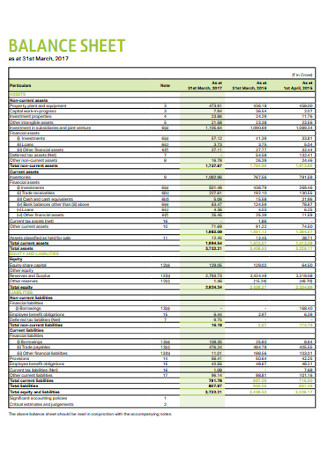
Balance Sheet Format
download now -
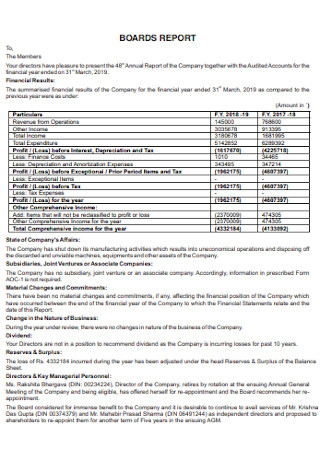
Board Balance Sheet Template
download now -

Business Balance Sheet
download now -

Model Balance Sheet Example
download now -
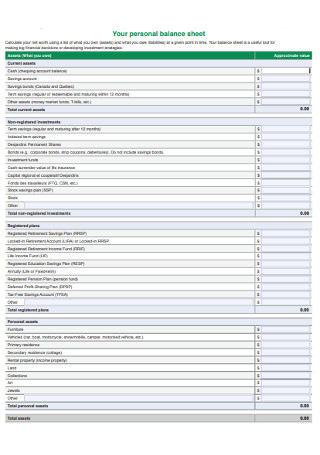
Personal Balance Sheet Template
download now -
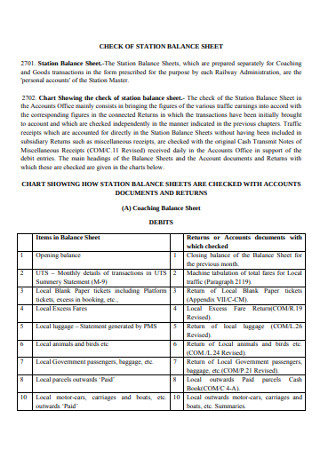
Station Balance Sheet
download now -
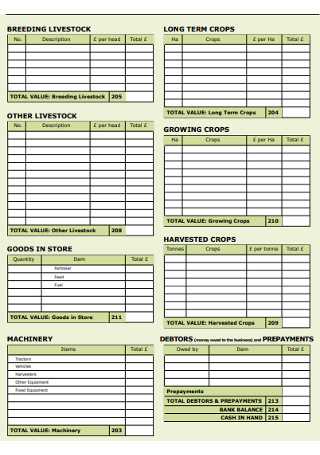
Former’s Balance Sheet
download now -
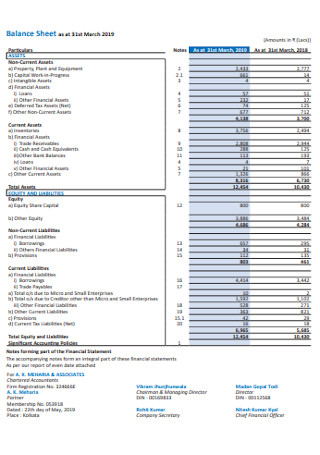
Balance Sheet Example
download now -

Liquidity and Off-Balance Sheet
download now -
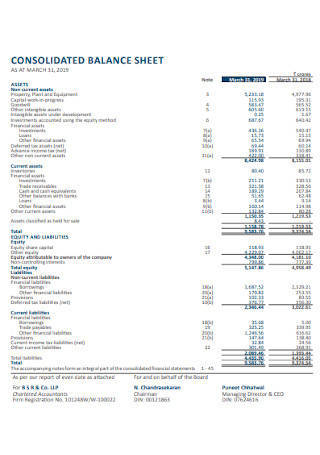
Consolidated Balance Sheet Template
download now -
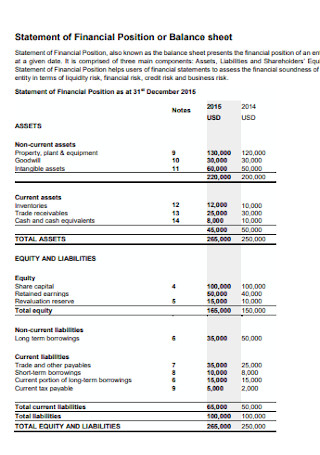
Financial Position or Balance Sheet
download now -
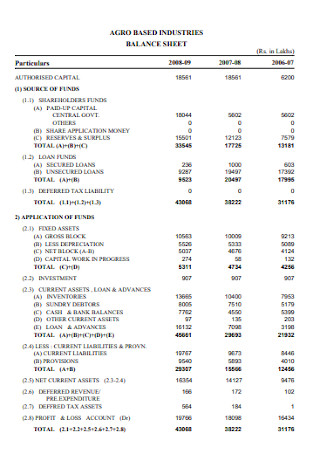
Industries Balance Sheet
download now -

Cash Flow Balance Sheet
download now -
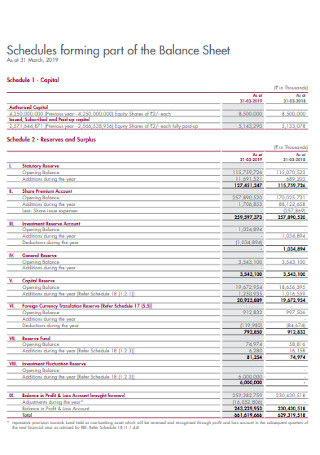
Schedules Forming Part of Balance Sheet
download now -
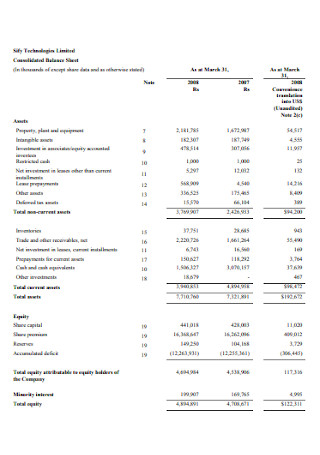
Consolidated Balance Sheet Example
download now -

University Balance Sheet
download now -

Accounting Balance Sheet
download now -

Personal Financial Balance Sheet
download now -
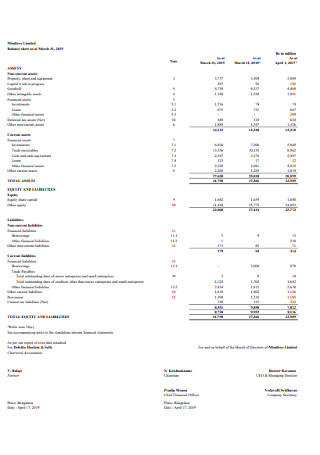
Basic Balance Sheet Template
download now -
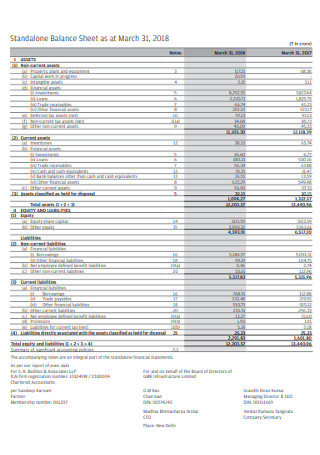
Standalone Balance Sheet
download now -
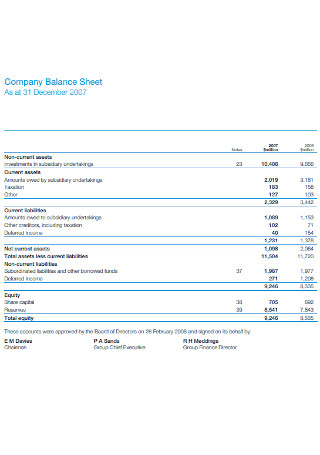
Company Balance Sheet
download now -
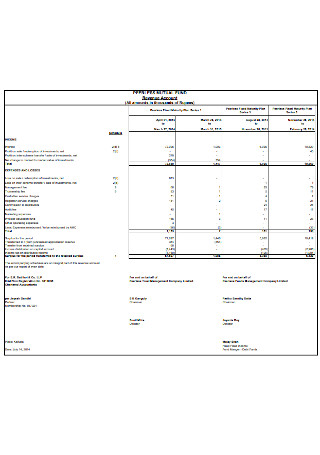
Mutual Fund Balance Sheet
download now -
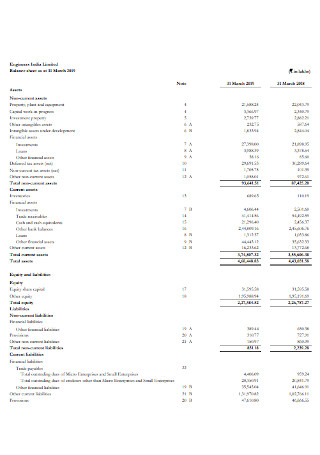
Engineers Balance sheet
download now -

Balance Sheets of Rural Banks
download now -
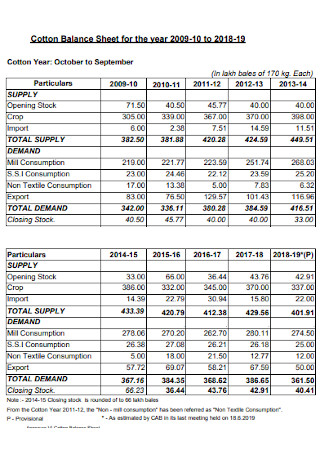
Cotton Balance Sheet
download now -

Cofeelab Balance Sheet
download now -
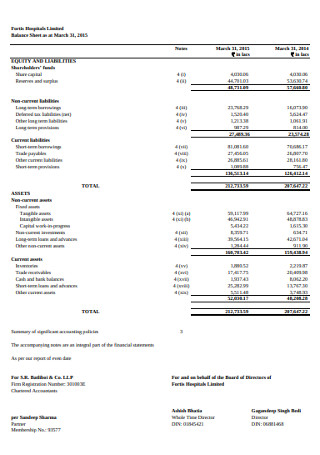
Hospital Balance Sheet
download now -
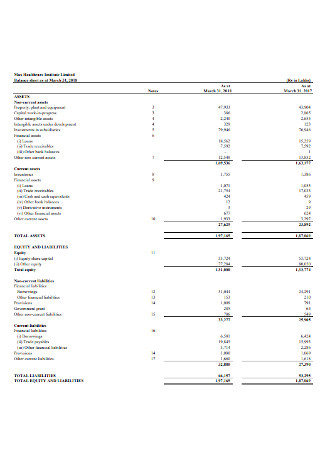
Healthcare Institute Balance sheet
download now -

Schedules to Balance Sheet
download now -
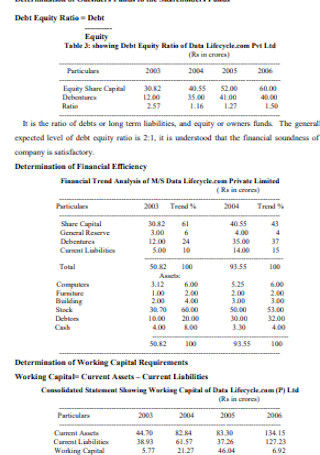
Study on Balance Sheet Analysis Template
download now -
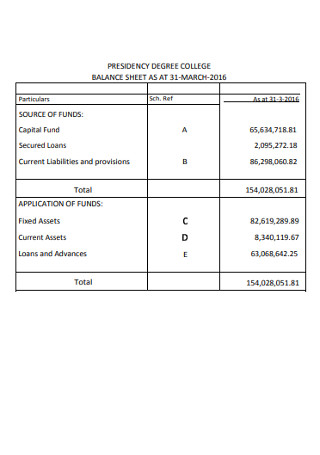
College Balance Sheet
download now -
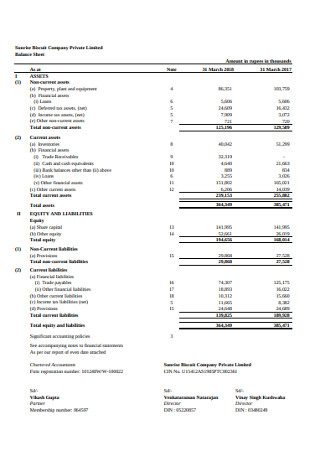
Biscuit Company Balance Sheet
download now -
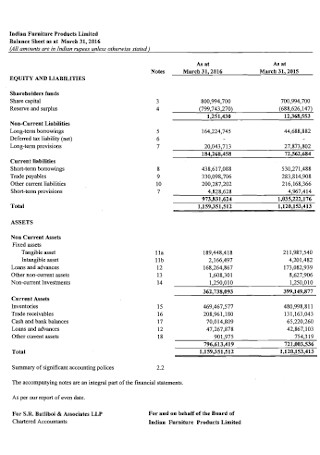
Furniture Balance Sheet
download now -

Verification of Balance Sheet Template
download now
What Is a Balance Sheet?
A balance sheet summarizes three factors—shareholders’ equity, liabilities, and assets. And this financial statement is useful to monitor and compute your organization’s capital structure, cash flow, financial health, and other data. For example, you refer to balance sheets in finding out how much money is left, if all assets have actually been sold, or if every debt has been paid back.
According to Investopedia, the standard formula for the balance sheet is Liabilities + Shareholders’ Equity = Assets.
The Three Main Sections of a Balance Sheet
A balance sheet’s basic formula is to add both liabilities and shareholders’ equity on one side and assets on the other side. But what exactly are these three factors? In this section, take a closer look at each balance sheet section and what they really mean.
How to Create a Balance Sheet
In the accounting sector, balance sheets play a great role in tracking finances and other data. But it hardly becomes a good reference when the balance sheet’s content is poorly made. So to ensure you come up with a proper balance sheet, kindly follow these steps:
Step 1: Set Your Balance Sheet’s Purpose
Every business has different objectives and purposes. So what about your organization? Focus on aspects related to what concerns the balance sheet like the income statement, cash flow, etc. Besides making a balance sheet for business, some people even create it for personal reasons. It really depends on your purpose and be sure to tailor it into your sheet.
Step 2: Identify the Assets, Liabilities, and Shareholders’ Equity
Of course, it is standard to insert the three sections of a balance sheet which are the shareholders’ equity, assets, and liabilities. You can conduct an analysis report towards your business first to identify those factors. Be careful with the numbers and data because some details are classified and you wouldn’t want to leak some. Also, it works best to be in partnership with other people in your team to work on this part quickly and effortlessly.
Step 3: Observe an Easy-to-Follow Format
You can freely decide how the format and design work for your balance sheet. The sample balance sheets listed above even have customizable features so you can change the details anytime. But, don’t overdesign the document where it ends up hard to follow already. Something simple yet understandable will already suffice. That way, you won’t have a difficult time understanding the content and how to use it.
Step 4: Update the Content Regularly
Making a balance sheet isn’t just a one-time process. Expect regular updates too that you may add, subtract, or alter the balance sheet daily, weekly, monthly, or anytime. And that regular assessment or monitoring is why you can keep track of whatever your company owes or owns. Aim to cover all important details from time to time and the balance sheet won’t fail you for sure.
FAQs
Who uses balance sheets?
Besides the accounting department, anyone concerned about a business’s financial health can use the balance sheet. This includes the investors, analysts, executives, regulators, and more.
What are the different types of assets?
The three types of assets are:
- Current assets (assets in the business for a short period or less than a year)
- Fixed assets (noncurrent and capital assets)
- Intangible assets (assets that can’t be touched like intellectual properties)
What are the types of liabilities?
Liabilities can either be short-term or long-term. They could all refer to loans, bills, charges, and other things a business owes. But the only difference is the duration. Short-term liabilities are payable in less than 12 months while long-term liabilities are payable in more than a year.
Using a balance sheet, you now have the official graphical data to state your organization’s different finances. Without it, you would have a hard time tracking your current assets, liabilities, and so forth. And failing to track them from time to time might have your business struggling financially. Thankfully, tracking has not been made easier with sample balance sheet templates up for grabs above. Download now!
