12+ SAMPLE Quality Audit Checklist
-

Quality Audit Checklist
download now -

Air Quality Self-Audit Checklist
download now -

Vendor Qualification Quality Audit Checklist
download now -
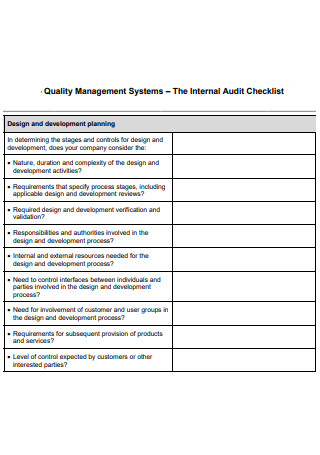
Quality Management Systems Audit Checklist
download now -

Sample Quality Audit Checklist
download now -
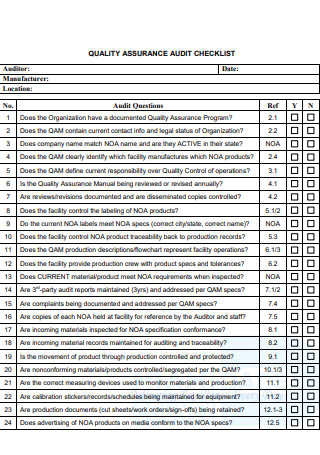
Quality Assurance Audit Checklist
download now -

Quality Control Audit In-Plant Checklist
download now -
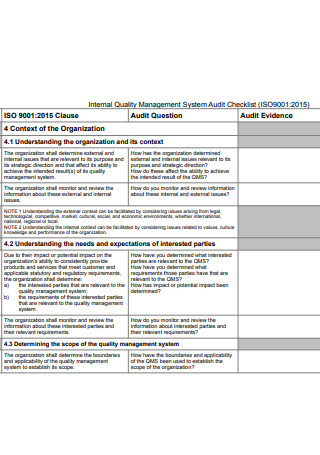
Internal Quality Management System Audit Checklist
download now -
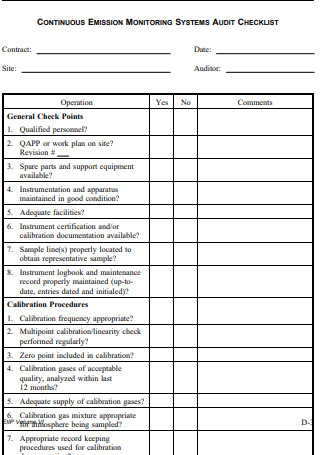
Data Quality Audit Checklist
download now -

Quality Audit Inspection Checklist
download now -

Internal Quality Audit Checklist
download now -
Sample Internal Quality Audit Checklist
download now -

Formal Quality Audit Checklist
download now
FREE Quality Audit Checklist s to Download
12+ SAMPLE Quality Audit Checklist
a Quality Audit?
Benefits of Quality Audits
Types of Quality Audits
How to Conduct an Internal Quality Audit
FAQs
What is a quality auditor’s role?
What is a quality management system checklist?
Do you double-check your checklist?
What purpose does a checklist serve?
What Is a Quality Audit?
Quality auditing is the systematic, independent, and recorded examination and evaluation of an institution’s quality management system (QMS) to specify whether quality activities and results conform to a strategic arrangement that is effectively implemented and appropriate for achieving the organization’s objectives. A quality audit is often undertaken at predetermined intervals by an internal or external quality auditor or audit team to ensure a business has a well-defined quality monitoring system. It is a critical component of the ISO 9001 quality management system standard. According to the Institute of Internal Auditors, confidence levels typically range between 90% and 95%. The word “confidence level” refers to an auditor’s degree of assurance that the sample will accurately reflect the population’s actual values. The greater the needed level of confidence, the larger the sample size.
Benefits of Quality Audits
Each organization has written and documented policies and standards. Thus, does every business prioritize internal quality standards first? However, is everyone genuinely concerned about the importance of quality reviews? “Quality” is a broad concept. The quality of organizational procedures has a more significant impact on functional areas than we may realize. In today’s world, corporations strive to enforce internal audits holistically. The pertinent question is “why?” Why do we require monthly internal audits? Do we need to repeat the same technique regularly? If you’re still interested, here are the answers.
Types of Quality Audits
At the most fundamental level, quality audits elicit objective proof of operational conformity. There are numerous sorts of audits that can be undertaken to determine conformance. Audits might be conducted internally, by a supplier, or by an independent third-party auditor. Audits can cover processes, systems, or products, but they are always compared to a predefined set of criteria. The following is a list of ten types of quality audits that are frequently performed:
How to Conduct an Internal Quality Audit
Few businesses recognize the value of internal quality audits. Auditing is frequently regarded as an undesirable but essential disruption. This requires time and effort and may cause employees to feel intimidated. And, once accomplished, minor changes. This is unfortunate, as internal audits are highly effective instruments. They can assist organizations in preparing for regulatory audits and external audits in identifying natural areas for development. Follow these six fundamental steps to conduct more effective internal quality audits.
-
1. Conduct routine audits
An audit should not be unexpected. Internal audits should be conducted regularly. How frequently should it depend on the type of your firm and the risk level associated with its activities? Additionally, avoid scheduling audits during peak work hours. This can result in unnecessarily high levels of tension and resentment. Include dates for internal quality audits in the annual calendar of the organization. Send reminders to staff as the audit date approaches. This allows individuals to prepare by gathering documentation, preparing for interviews, and verifying their bookkeeping.
2. Define the audit’s scope
Typically, in cooperation with team experts, the quality manager is most equipped to develop internal audit criteria. These should be concentrated on high-risk regions throughout the process or company lifecycle. They should also include the entire workflow to discover inefficiencies. It is critical to keep consistency in the criteria across time to the extent that this is possible and makes sense. This provides team members with specific objectives to work toward between audits. Additionally, it enables the evaluation of performance over time. However, criteria must be adaptable to your organization’s growing procedures.
3. Prepare for the audit
An internal quality audit must be planned before it can happen. You will need to hire an auditor. This could be an employee who knows about the process but isn’t directly involved. It could also be someone who is not from the company but knows about business and industry rules. Employees from different departments usually help with the audit. Suppose they see what will be audited and how the process is less scary. Set the format for the audit. The interviews could be about the process, the documentation, or the workflows. This could also include a demonstration of how the workflows work. A good audit includes clear communication about what the audit is about and how it will be done. Another thing that can help is if employees have a set amount of time to prepare for the audit.
4. Perform the audit
To guarantee maximum participation and cooperation, begin by convening a meeting to inform staff about the audit’s objectives. Additionally, work to dispel worries of a witch hunt targeting underperforming team members. Encourage participants to submit their observations and enhancement suggestions. Allow sufficient time for the auditor to obtain a thorough understanding of your company’s business life cycle and workflows once the auditing process begins. Additionally, it is beneficial to analyze the organization’s quality management system to discover any irregularities.
5. Report on the audit
The auditor’s final report lays out the audit findings and recommends what to do next. It should help managers keep track of quality and performance over time and look for ways to improve. Often, these reports aren’t shown to anyone else in the company. Instead, it should be communicated to the workforce in a summary form with clear areas for improvement. At the same time, point out how well the team has worked since the last review. This can help keep workers excited.
6. Take appropriate action on recommendations
This is the critical phase. Demonstrate to employees that quality and the auditing process are important by acting on the findings. Concentrate on one or two enhancements at a time. Conduct regular evaluations of the outcomes of implemented modifications and their impact. Additionally, strive to involve employees in resolving the auditor’s recommendations. Everyone must take an active role in any effort to develop continuously. Additionally, it is critical for maintaining long-term compliance with quality standards.
FAQs
What is a quality auditor’s role?
Quality auditors keep an eye on the products and services of a business. They define parameters for testing products and services, devise quality control methods, conduct audits, and supervise quality control teams. They may work in various industries, including manufacturing and supply, food & beverage, and information technology.
What is a quality management system checklist?
A quality management system is a software application used to formally document processes and procedures inside a company. Having defined processes and procedures enables a business to benchmark and measure its quality and give metrics to improve the business’s quality.
Do you double-check your checklist?
When team members utilize a Do-Confirm checklist, they accomplish their tasks from memory. Then they pause and go over the list, confirming that they completed each thing. When using a Read-Do list, individuals do each action as they check them off, much like a recipe.
What purpose does a checklist serve?
Checklists detail each stage of a procedure, which helps keep things organized. It can be used as a visual reminder, a method for prioritizing chore lists, and a means to schedule all that has to be done to avoid missing deadlines. Simple to use and highly effective at ensuring that all procedures are completed.
Quality auditing is an effective management tool for determining the root reasons for poor quality, corrective actions, and quality-related confirmation or verification activities. It identifies quality management system flaws and errors and assists in improving and resolving them based on comprehensive findings and recommendations. Are you prepared to build your quality audit checklist based on the above facts? If you are, there are downloadable templates accessible for you!
